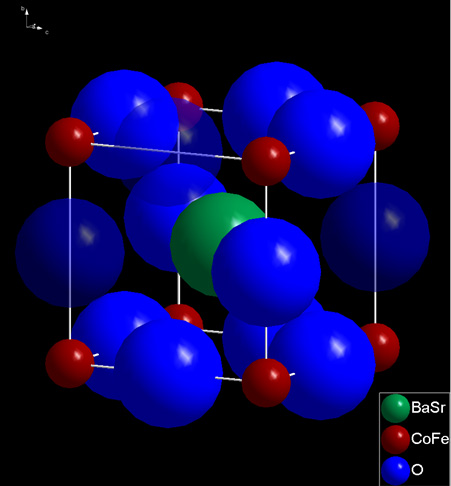
The development of mixed ionic and electronic conducting membranes requires a fundamental knowledge on the physicochemical characteristics of the materials used. The oxygen transport is realized by the oxygen vacancies inside the oxygen sublattice of the crystal structure. Up to a quarter of the regular oxide ion sites can be unoccupied. The majority of application-relevant properties depends on the concentration of these oxygen vacancies.
Services offered
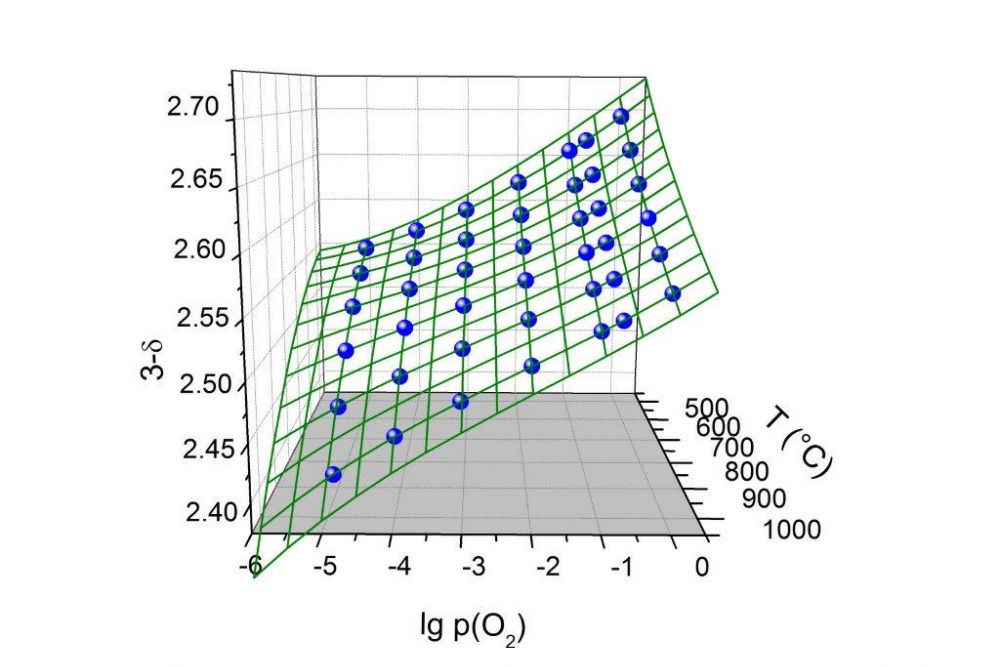
- Characterization of the dependence of the oxygen stoichiometry on temperature und oxygen partial pressure by highly sensitive thermogravimetry at oxygen partial pressures from 1 bar to 10E-20 bar
- Creation and parameterization of defect models
- Determination of phase boundaries and phase composition (XRD/Rietveld)
- Determination of the thermal and chemical expansion coefficients by dilatometry and XRD
- Determination of the absolute oxygen stoichiometry by cerimetric titration
- Determination of the reaction enthalpy of the oxygen absorption and desorption (TG/DTA and DSC)
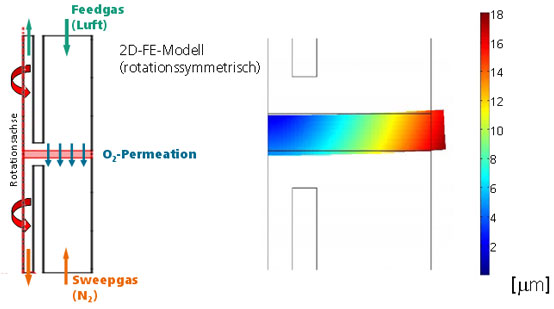
The research occurs in cooperation with the Fachhochschule Jena and the Bauhaus-Universität Weimar. The results makes it possible to simulate the mechanical stress situation inside an oxygen permeable membrane and also the O2 permeation of membranes and other ceramic components at application conditions.