
Lab-scale demonstration of an integrated Co-electrolysis-based power-to-X process
Current research



When it comes to mitigating industrial CO2 emissions, power-to-X processes are frequently discussed as a technological option for utilizing these emissions. In particular, high-temperature electrolysis (solid oxide electrolysis, SOE) is a promising technology. It efficiently converts CO2 and water into syngas, and subsequently into valuable hydrocarbon products.
High-temperature electrolysis-based powerto-X plants can reach high process efficiencies, because the cathode is catalytically active for reforming short-chained by-products of the synthesis step (e.g. methane). This allows for the direct conversion of these low-value products back into syngas, leading to increased carbon efficiency,
ηC1, products
ηC= ___________
ηCO2, input
Moreover, it reduces the complexity of the entire process, since no additional reforming step has to be included.
In order to demonstrate the potential of internal reforming within an integrated process, a power-to-liquid lab-scale plant, consisting of high-temperature co-electrolysis coupled with Fischer-Tropsch synthesis, was implemented as part of the project “Colyssy” (FK 03ZZ0741A), funded by the German Federal Ministry of Education and Research (BMBF). The rated electric input into the electrolyzer was Pel,SOEL = 1 kW. For the fixed-bed Fischer-Tropsch reactor, an industrial cobalt catalyst was chosen for its performance and favorable product spectrum, yielding high-value long-chained hydrocarbons. The lab-scale plant was successfully operated for over 1200 hours. Over the course of the experiments, different operating conditions of the electrolyzer and the Fischer-Tropsch reactor were tested.
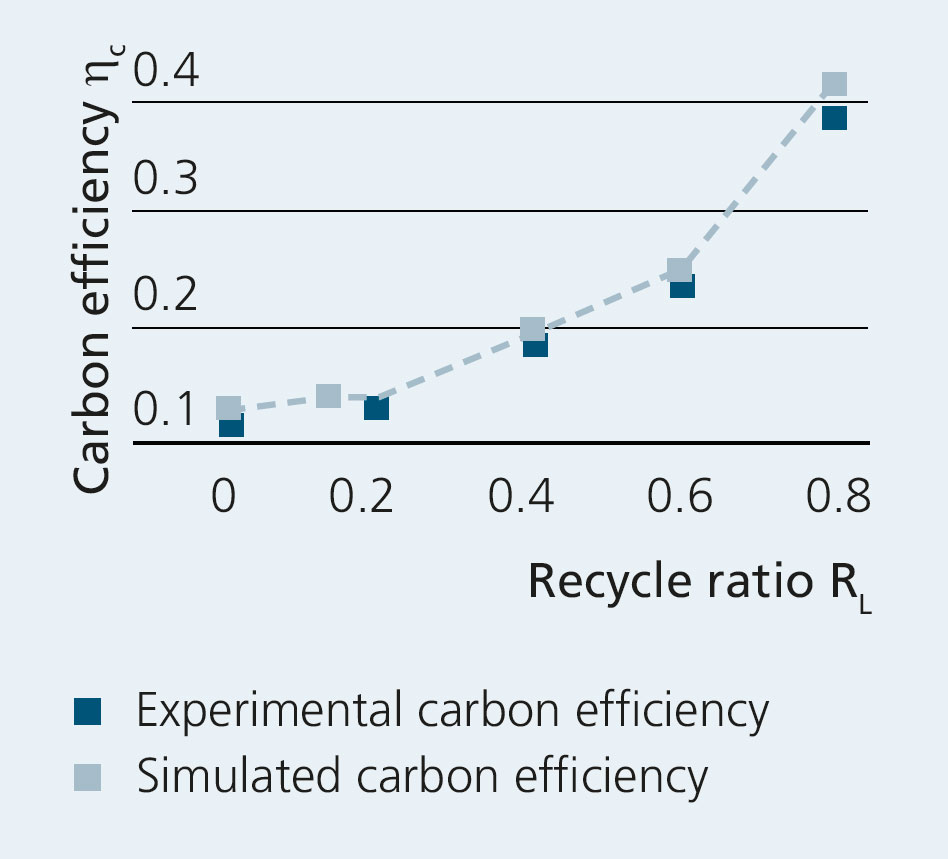
A special focus was placed on the variation of the recycle ratio,
mtail gas
RL= _______________
mtail gas + moff-gas
which represents the fraction of the Fischer-Tropsch product gas stream recycled into the electrolyzer. The aim of this variation was to demonstrate the potential increase in carbon efficiency ηC through by-product utilization via internal reforming, and to highlight the inherent advantages of high-temperature electrolysis.
The results of the tests have shown a significant increase in carbon efficiency as the recycle ratio increases, without the electrolyzer showing any sign of performance degradation. The data were correlated with a process model and a good agreement between experiment and simulation was found. Increasing the recycle ratio RL further – above the value of 0.8 adjustable for the lab-scale plant – would be possible on a larger scale, and modeled data suggest a significant further increase in carbon efficiency ηC.
All knowledge derived from the lab plant and the process model will be used to upscale the technology. In this context, a container-based plant will be realized at a lime production facility mutilizing CO2 derived from exhaust gas. Additionally, possible means of heat integration will be investigated.
Supported by

