
Titanium oxides – electrical all-rounders
Current research

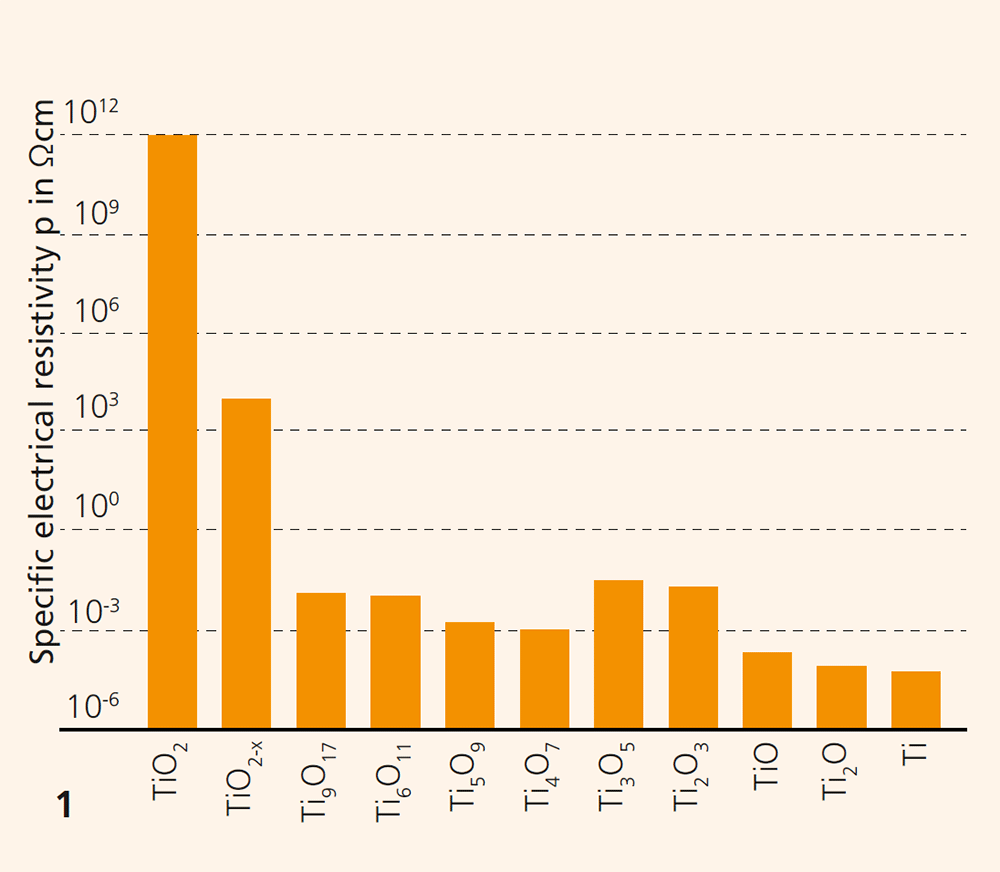

At Fraunhofer IKTS, titanium oxides are adapted to customer-specific user requirements, components are manufactured from them and extensively tested. Titanium oxides are characterized by extraordinary variability concerning their oxide compounds. Besides titanium dioxide (TiO2), which is used for numerous products, such as wall paints, cosmetic products, paper or carriers of catalysts, there are multiple other oxide compositions of titanium. Their variable compositions range from Ti2O to TiO2. Even a tiny oxygen deficiency in TiO2 can decrease the electrical resistance from 1012 Ωcm to a level between 105 and 103 Ωcm. The crystallographic phase change to e.g. Ti4O7 shifts the electrical resistance to 10-2–10-3 Ωcm. Figure 1 illustrates the proportions across 15 orders of magnitude of electrical resistivity covered by various titanium oxide types. Furthermore, non-linear correlation of voltage and currency can be created through the doping of TiO2. Electrical permittivity is particularly high, at a level between 60–800 depending on the applied frequency. Significant oxygen conductivity already sets in at 500 °C. Also of technical interest is the photocatalytic effect, which is particularly linked with the TiO2 type anatase.
Electro-technical applications
This wide range of properties offers advantages in numerous technical applications: specific materials for electrodes, thermo-electric materials, varistor materials, active catalyst substances, electrical conductors, semi-conductors and insulators or even sensors for oxygen identification and analysis. These applications have a variety of different requirements regarding densification, mechanical strength or oxidation stability. Through the specific modification of the manufacturing parameters, titanium oxide ceramics can be produced as dense and high-strength material or made to be porous and gas-permeable. TiO2 is completely oxidation-resistant and can be used up to 1500 °C under air. Suboxides are oxidation-resistant up to 400 °C. Furthermore, titanium oxides are chemically stable against almost all reactive substances.
The electrical parameters can be modified to suit almost all applications. Joined compounds of titanium dioxide and titanium suboxides can be produced which combine electrically insulating and electrically conductive parts within a monolithic component (Figure 2).
Services offered
- Adaption of titanium oxides to specific application requirements
- Manufacture of customer-specific titanium oxide components
- Implementation of material- and application-orientated investigations and tests