Highly active nanostructured TiO2 filtration membranes for water disinfection and elimination of trace substances

Clean drinking water is one of the most precious commodities of our modern civilization, but ensuring its supply as the world’s population is growing is a great challenge. The constant input of fertilizers, chemicals or medication residues into our waters and the associated presence of anthropogenic trace substances that are difficult to remove are damaging our available water resources more and more. Photocatalysis is a promising process for purifying water. It enables the complete degradation of organic contaminants without producing residual substances that need to be disposed of. However, solid particles cannot be removed.
A novel approach to comprehensive water purification developed by Fraunhofer IKTS consists in combining a ceramic filtration membrane with a photocatalytically active layer. A porous filtration membrane made of glass-bonded silicon carbide serves as the carrier structure. This is mechanically very stable and has excellent abrasion and corrosion resistance. Titanium dioxide (TiO2), as photocatalyst, is applied in the form of a nanostructured layer on the surface of the ceramic membrane. This nanostructuring creates a large photocatalytically active surface area.
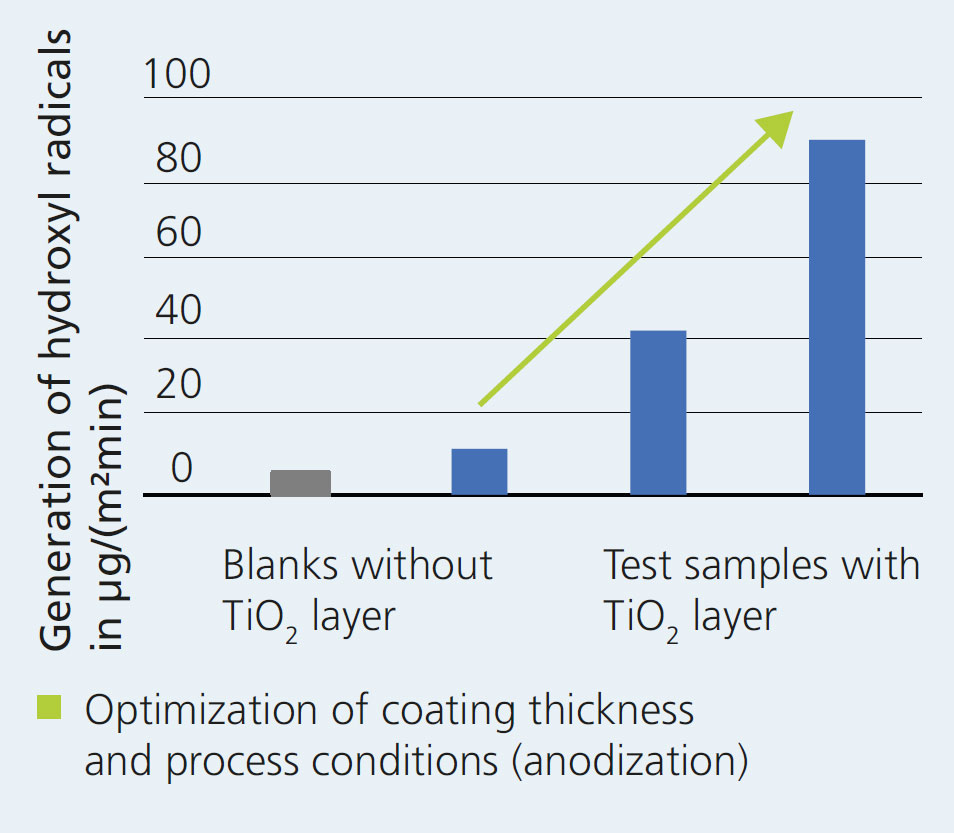
The ceramic membranes are photocatalytically coated in a two-step process. First, pure titanium is deposited on the ceramic surface by means of the technically very simple and easily scalable magnetron sputtering process. This allows the economically efficient creation of layers with thicknesses between a few nanometers and several micrometers, even on largearea substrates. Subsequently, the dense titanium layers are converted into TiO2 layers with ordered nanoporosity by means of the technically established process of anodic oxidation, in which closed or porous oxide layers with adjustable morphologies (e.g. pore widths and lengths) are produced by selecting the anodization parameters in a targeted way.
The photocatalytic effectiveness of the nanostructured titanium dioxide layers produced in this way has already been successfully demonstrated in the laboratory. The short-wave UV light irradiation of the TiO2 induces the formation of highly reactive hydroxyl radicals which break down contaminants oxidatively with high efficiency.
The work for this topic was supported by the Deutsche Bundesstiftung Umwelt (DBU) and carried out in cooperation with the company scia Systems GmbH.
Supported by