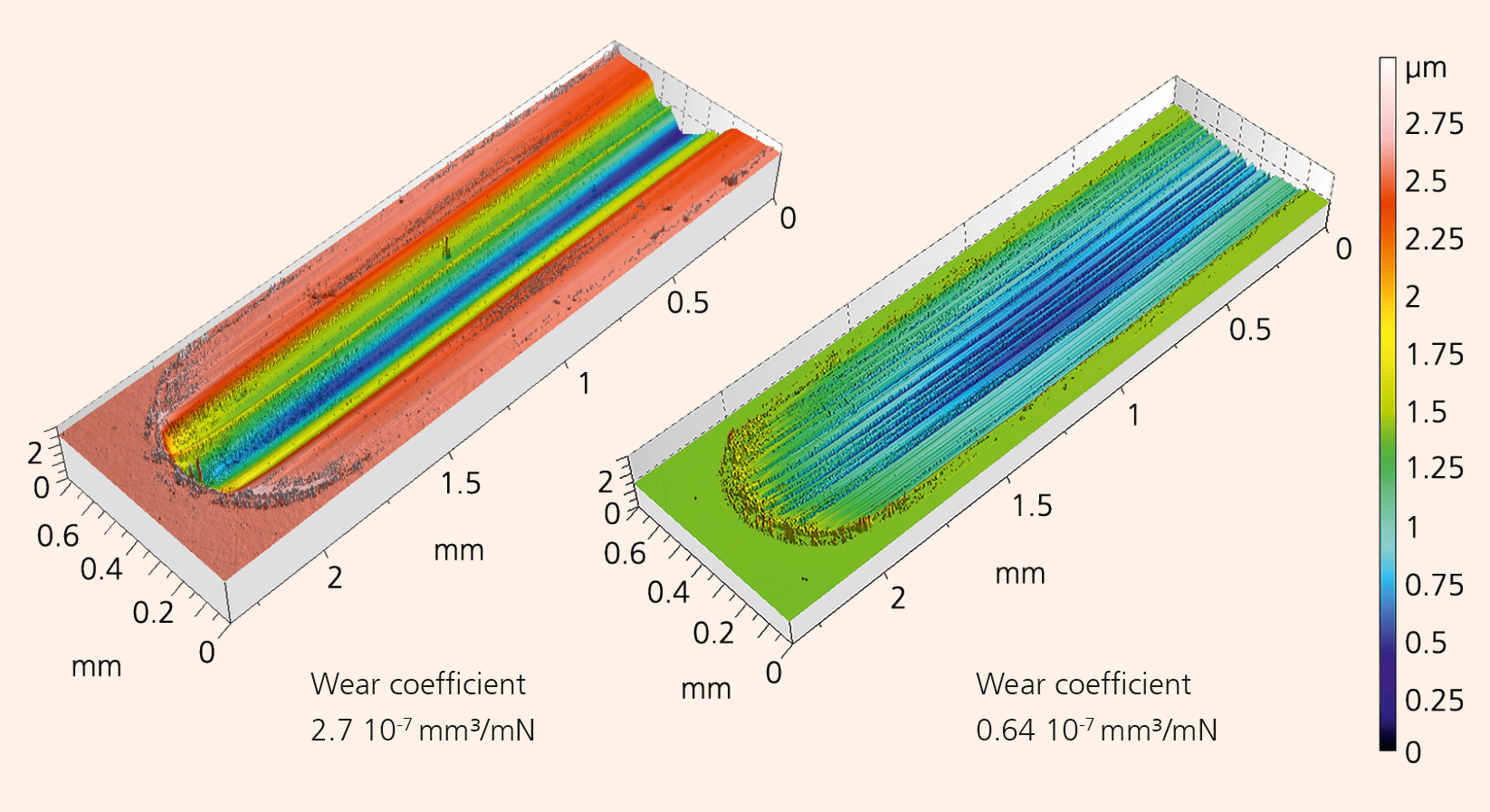

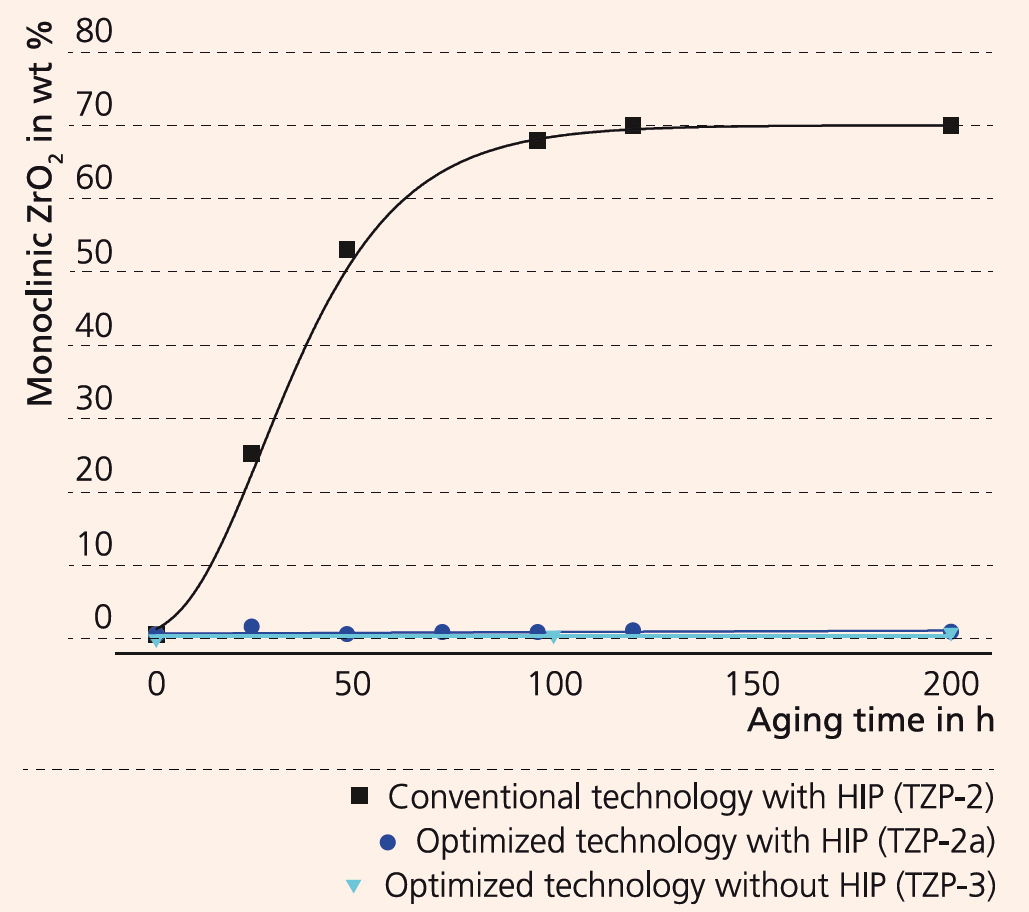
At Fraunhofer IKTS, there is a wealth of experience in the area of raw material dispersion and shaping. Using cost-effective commercial submicron-powders, it was made possible to create a ceramic material which combines increased translucency (Figure 2), hydrothermal resistance (diagram), high hardness, and wear resistance (Figure 1): tetragonal yttria-stabilized zirconia (TZP). Such ceramics are ideal for dental implants, but also for industrial applications. Analysis has shown that translucency of TZP can be achieved by reducing the grain size of the microstructure and by reducing residual porosity and impurities. This is confirmed by measurements of the in-line transmission of TZP ceramics with different mean grain sizes in the microstructure (TZP-2: 340 ± 30 nm, TZP-2a: 150 ± 30 nm). These ceramics were post-densified by hot isostatic pressing (HIP). A reduction of the grain size by 55 % doubles the translucence. Even without HIP (TZP-3), there is a visible increase in translucence at an average particle size of 190 ± 10 nm. Furthermore, hydrothermal aging was simulated for 200 hours at 134 °C and 2 bar in the autoclave. The diagram shows the subsequent phase composition. While sample TZP-2 showed a rapid increase of the monoclinic phase, samples TZP-2a and TZP3 presented no increase of the monoclinic phase, are therefore hydrothermally stable. Additionally, interference microscopic images of wear tests (Figure 1) showed that fine grained TZP-ceramics are four times as wear-resistant as coarse-grained probes.
Services offered
- Development of preparation and shaping technologies for oxide ceramic materials
- Development of prototyped
- Certification according to ISO 13485 (quality management for medical devices)