
Hybrid and degradable bioceramics for jawbone replacement
Current research



Bone defects in the craniofacial region are often associated with esthetic and functional impairments. Until now, autologous bone grafts, e.g. from the fibula or the pelvic bone, have been used for reconstruction. However, these offer only an inadequate reproduction of the complex anatomy of the facial skull.
New approach for bone regeneration
The “Hybrid-Bone” project (04/2020–03/2023,funding code 03VP07633) aims to develop personalizable and precisely fitting, mechanically stable bone substitute materials. The combination with growth factors in the degradable part of a hybrid scaffold also stimulates programmable bone regeneration. The hybrid bone consists of additively manufactured, load-bearing support structures filled and sorrounded with porous foam. The foam serves as a biodegradable lead structure for cell ingrowth.
Promising material combinations
The bioceramic materials used are degradable tricalcium phosphate (TCP) and bioinert zirconia. The CerAM VPP (Ceramic Additive Manufacturing Vat Photopolymerization) process was selected for additive manufacturing and fabrication of the pressure-loadable support structures from a TCP-ZrO2 composite material. Specifically constructed holes were provided for subsequent screw fixation in the bone (top image, implant size 20 x 12 mm; H x ø). The ceramic foam was prepared from TCP via the so-called Freeze-Foaming process. Such a TCP Freeze Foam has open and interconnected pores. This allows it to be degraded by the body and at the same time cause new bone formation at the same location.
Good biocompatibility has been demonstrated on the hybrid test scaffolds based on in-vitro analysis: the middle image shows living primary human osteoblasts in the hybrid bone. Gene expression data of the primary osteoblasts, cultured on different materials, show when compared to the reference (Bio-Oss® Spongiosa Blocks, Geistlich Pharma AG) that the osteogenic markers Runx2 and COL1A1 are expressed equivalent to the reference.
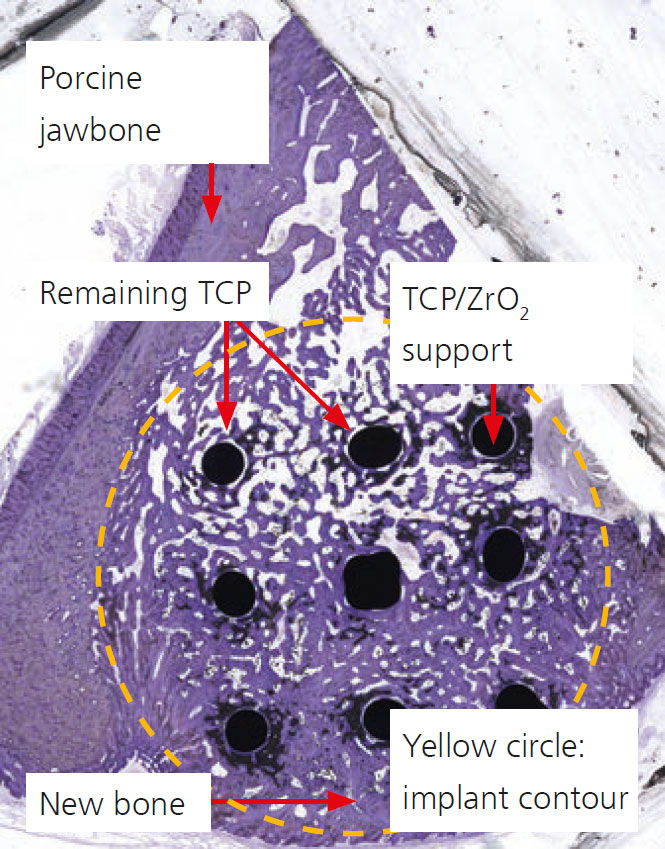
Results of the first in-vivo test showed that the artificial porous scaffold was degraded within only three months, and autologous bone was newly formed at the same location (bottom figure).
With these outstanding biocompatible properties, the bone substitutes will be developed further to take into account the natural distribution of loading forces in the respective bone while paying more attention to biomechanics. Chewing and muscular forces play a major role in the constant formation and degradation of bone tissue. Currently, larger hybrid bones (approx. 50 x 50 x 20 mm³) adapted to porcine jaw bones are being prepared and will subsequently be evaluated in vitro and in vivo.
The goal at the end of the project is to produce a personalized human jawbone for potential use in patients.