
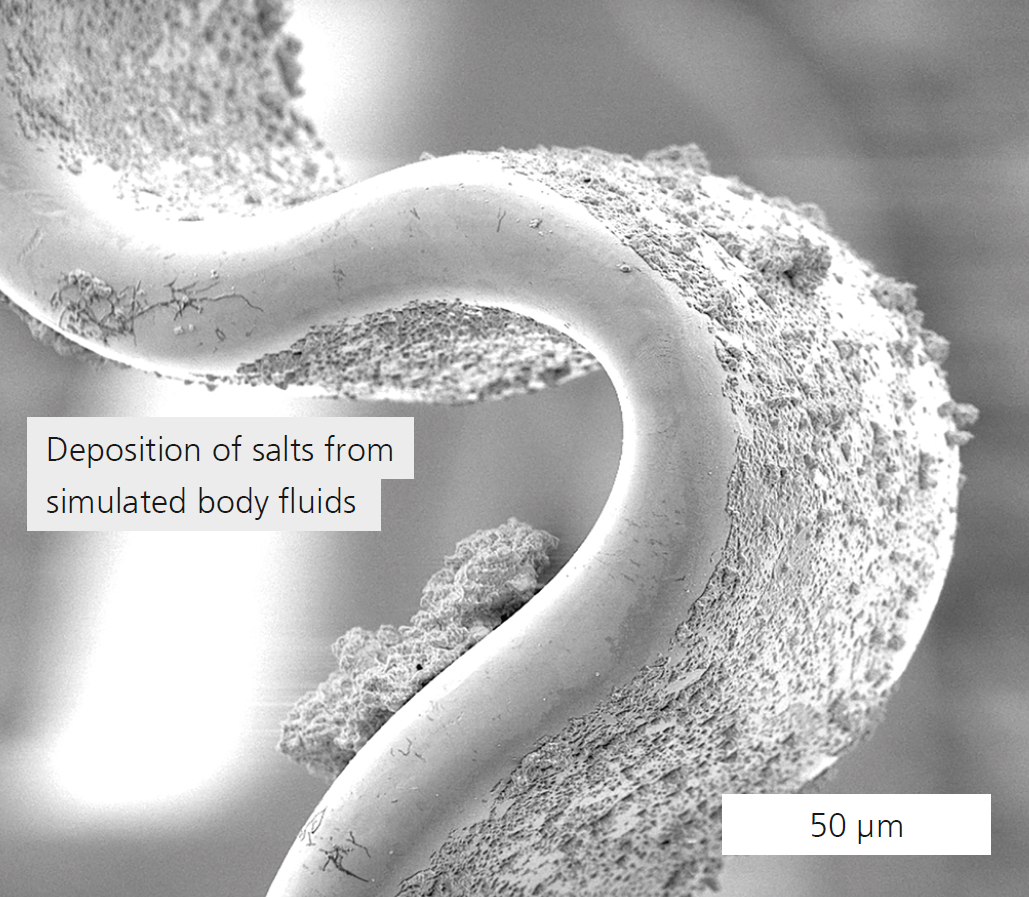
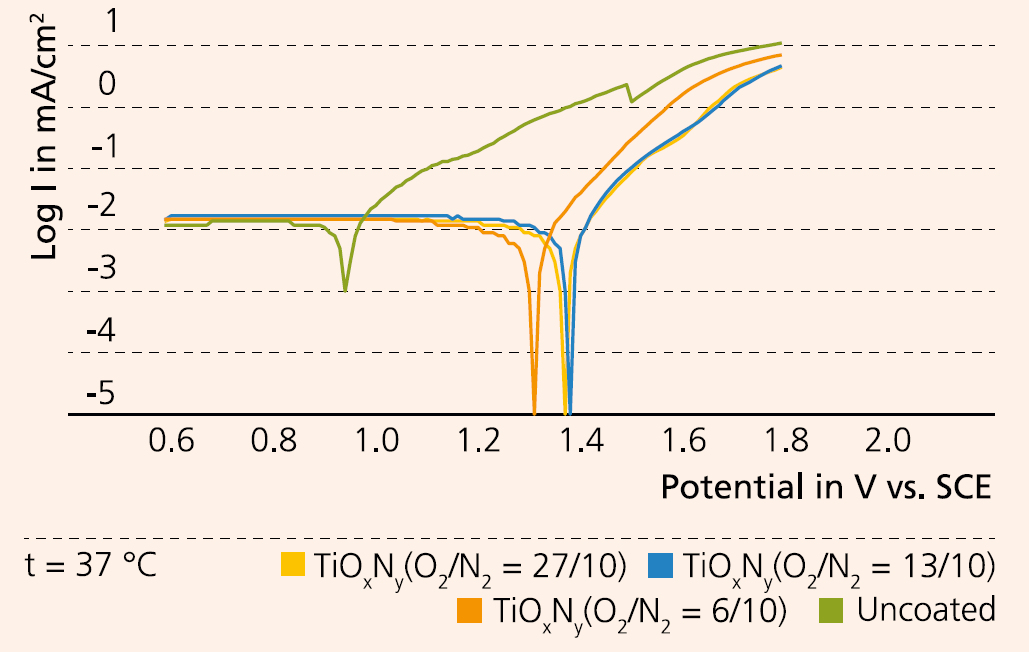
Heart disease is the leading cause of death worldwide. Stenting of arteries is a common treatment to open up constricted or blocked blood vessels. Even though the risk of complications is low, restenosis (the re-narrowing of arteries) remains a clinical problem, and stents are coated to prevent it. One promising stent coating types is titanium oxynitride (TiOxNy), deposited by magnetron sputtering. Its biocompatibility improves with increasing thickness of the coating film. But by increasing the film thickness, its adhesion to the substrate deteriorates drastically, causing coating defects (Figure 1). The research project “TiOxTechBio” (Funding code: 01DJ15023) led by Fraunhofer IKTS with partners from Poland, Romania, and Russia, has developed a new technology enabeling the manufacture of TiOxNy coatings for cardiovascular stents made of stainless steel (316L) with low thickness, high hardness, high corrosion, wear and oxidation resistance and sufficient biocompatibility. IKTS has contributed to the joint project by establishing an in-vitro method to investigate the long-term stability of different coated stent prototypes and by carrying out studies of physical-chemical surface properties and corrosion tests. Two determining factors for the TiOxNy coating microstructure were confirmed by the experiments: the nitrogen concentration in the gas atmosphere of the vacuum chamber and the supply of the negative bias to the substrate. We were able to determine an optimal O2/N2-ratio of 3/5 and an optimal coating thickness of 150 to 170 μm. An increase of the nitrogen concentration leads to a reduction of the grain size, increasing surface roughness and enhancing the coating tightness. The development of the deposition technology focused on improving coating quality, flexibility, and on achieving a good adhesion. These goals were reached by optimizing the deposition parameters in several steps by technological means, and through dynamic testing with simulated body fluids (Figure 2). Analysis of the test fluids after their interaction with stent surfaces of optimal design by means of mass spectroscopy with inductive coupled plasma showed no release of the coating elements, confirming the good stability of the surfaces. After successful in-vivo and clinical studies, the deposition technology developed so far can be transferred to industrial stent manufacturing.
